The technique of applying a voltage to a gas to produce a glow discharge, or "plasma" technology, has become a powerful tool for solving surface pretreatment problems in the field of medical devices. The plasma can be used not only for extreme cleaning and disinfection of surfaces, but also for improving the adhesion of biomaterials to in vitro diagnostic platforms and biocompatible coatings to in vivo instruments. Indeed, the plasma not only activates the surface to facilitate the fixation of cells or biomolecules, but can also in turn produce a smooth surface that resists biofouling or is used to meter the formulation of the drug. Plasma can also greatly enhance the efficacy of microfluidic devices. Microchannels on clinical diagnostic devices can become more "wet" to biological fluids without affecting their analytical performance. Plasma is also used in some low-end technologies, such as improving the ink marking of the catheter and increasing the adhesion of the syringe needle to the syringe. In addition, because plasma is a dry surface treatment technology that does not require the disposal of waste chemicals, it becomes a green process that requires very little consumables. In this article, we will discuss the capabilities of plasma technology in the in vitro diagnostic platform industry. We will focus on how the plasma controls the surface energy and how to modify the surface chemistry to improve adhesion to biological materials. What is the scientific basis behind the magical modification of the surface of the plasma?
What is a plasma?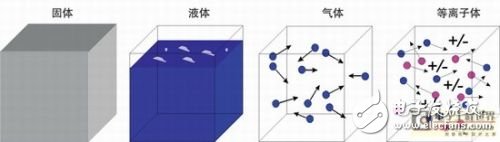
Figure 1: Schematic diagram of the four material states. The fundamental difference between a plasma state and a gaseous state is that the plasma state can be electrically conductive. The electrons get rid of the attraction of atoms or molecules and can transfer energy through the collision of electrons.
A plasma, like a solid, liquid, or gas, is a state of matter. Apply enough energy to the gas to ionize it into a plasma state. The "active" components of the plasma include: ions, electrons, reactive groups, excited states of nuclides (metastasis), photons, and the like. Controlling and controlling the aggregated properties of these active components can be subjected to a variety of surface treatments such as nanoscale cleaning, infiltration of activated surfaces, chemical grafting, coating deposition, and the like.
The high chemical activity of the plasma is used to alter the properties of the surface without affecting the substrate. In fact, it is possible to control the energy carried by the gases separated by these parts, so that they contain very low "hot" energy. The method is implemented by coupling energy to free electrons rather than to heavier ions, so that heat sensitive polymers such as polyethylene and polypropylene can be treated. How is energy coupled to the gas? In most cases, an electric field is applied between the two electrodes in a low pressure environment. This is like how a fluorescent light works. The only difference is that it doesn't let light shine. We dominate his chemical properties to handle the surface of the material. Plasma can also be produced at atmospheric pressure. In the past, atmospheric plasma temperatures were too high to be used as a tool for surface treatment. Recently, improved techniques have been able to generate low temperature plasmas at atmospheric pressure and are applicable to the processing of most temperature sensitive polymers.
How does plasma change the performance of the surface?
Figure 2: Plasma as a surface treatment tool is mostly produced in a low pressure vacuum chamber. With the advancement of technology, the generation of plasma under atmospheric pressure has begun to spread and is being used more and more. Figure 2a is a benchtop low pressure plasma system from PVA Tepla. This type of system has advanced performance and is well suited for use in the unit industry as well as in the laboratory. Figure 2b is a close-up of an atmospheric pressure plasma pen from PVATepla. This design safely controls the voltage and current inside the plasma pen body, which can be used for in-line applications or selective local processing.
Suppose a solid surface adsorbs hydrocarbon contaminants. These contaminants are easily reacted with plasma oxygen species. Oxygen attacks the adsorbed hydrocarbons, which are converted into CO2 and H2O. Figure 3 is a simple reaction mechanism. For easily oxidizable surfaces, surface cleaning with plasmad hydrogen is optional. Hydrogen can not only convert part of the organic matter on the surface into volatile hydrocarbons, but also reduce the oxidation of metals such as copper, nickel and silver.
The chemical properties of the plasma depend almost on the source gas. For example, O2, N2, N2O, CO2, etc. can generate an oxidative plasma. These gases are used to make the surface more infiltrated or polar to the polar solution. This is achieved by plasma-induced covalent oxygen bonding to a functional group such as a carbonyl group, a carboxyl group, or a hydroxyl group. These polar functional groups increase the energy of the surface and, therefore, allow tissue cells to adhere better or allow analytes dispensed onto the diagnostic platform to flow more easily through the microfluidic channels.
Ar/H2, NH3, etc. can generate a reducing plasma. These gases have been shown to be effective in activating fluorocarbons such as PTFE. Because of the inertness and biocompatibility of PTFE, it is an ideal material for the manufacture of medical devices in the body. However, these characteristics are also disadvantageous factors in the processing of PTFE, such as adhesion to synthetic scaffolds to promote tissue growth in the body device. The reducing plasma can solve these problems by lowering the fluorine concentration of the entire surface and replacing the fluorine atom with a functional group such as a hydroxyl group. The hydroxyl groups on the surface provide anchor points that support these synthetic scaffolds.
Some applications require erosion of the primary material. Fluorine-containing gases such as NF3, SF6, and CF4 are suitable for etching hydrocarbon polymers, silicon, and materials such as silicon oxide and silicon nitride. In addition to the strong chemical action of the plasma, the direct action also plays an important role. Particles with kinetic energy impact the surface to remove more surface inert contaminants (such as metal oxides and other inorganic pollutants). And cross-linking the polymer at the appropriate location to maintain the effect of the plasma treatment.
The polymer coating can be grown by a plasma enhanced chemical vapor deposition (PECVD) process. PECVD works by activating a nucleus such as a monomer in a plasma and inducing polymerization at the base of the workpiece. PECVD coatings have some properties such as protective layer, anti-sticking, and scratch resistance. In addition, some coatings contain some special functional groups such as -NH3, -OH, -COOH. These functional groups provide suitable bonding sites for subsequent grafting (eg, immobilization of proteins or sensing agents for biological materials), or can increase the binding of functional coatings (eg, anti-prothrombin, lubricated, type IV collagen) Wait). The surface chemistry of the deposited coating is determined by the outer surface at depths of tens of nanometers.
What can plasma do for the IVD platform?The application of plasma in the medical device industry is indeed very extensive. Therefore, this article will focus on application areas that have been validated by our R&D department and related to the medical diagnostic platform industry. In this field, plasma is used to prepare the surface for downstream processes, as well as to activate the surface to facilitate the bonding of biological materials. The latter is achieved by changing the surface polarity, grafting specific functional groups or polymerizing the coating on the surface. To better understand how the plasma adjusts the surface to meet the needs of the application, let's look at some important examples.
Microfluidic device and hydrophilicity
Surface energy is a material property that determines factors such as wettability and biofouling susceptibility. Generally, materials with high surface energy are hydrophilic and are wettable to fluids such as plasma, bacterial cell suspensions, buffers, inks, glues, and various adsorbents and coatings. On the other hand, a low-energy surface is called hydrophobic and has a "non-stick" property. These "non-stick" surfaces are discussed below.
Typically, microfluidic devices require a hydrophilic surface so that the analyte can continue to flow smoothly through the microchannels to the detection and processing components. This flow can be achieved by various methods of suction, electroosmosis, heat, mechanical, and the like. Like the medium (see below), the microfluidic device is made of a hydrophobic polymeric material (acrylic, polystyrene, polydimethylsiloxane (PDMS). A major problem caused by the hydrophobicity of these materials is The bubbles trapped in the microchannel suppress the flow of the liquid. Even if the channel is treated with alcohol and buffer, there is still a bubble problem. Plasma treatment can oxidize the surface of the microchannels, making them hydrophilic, thereby preventing The formation of bubbles. The surface charge density during electrokinetic pumping also affects the flow rate. Electrokinetic pumping drives the fluid through the microchannel by the principle of electrical reaction that converts electrical energy into kinetic energy. The charged surface attracts counter-productivity in the electrolyte. Charged particles. This allows the particles to remain in the fluid and pass through the channel more easily by electrokinetic suction. The plasma can effectively promote electrophoretic or electroosmotic flow of the charged surface.

Figure 3: The above reaction mechanism is a simple schematic diagram of a plasma-generated oxygen group attacking hydrocarbons adsorbed on the surface. There are numerous other mechanisms including different oxygen excited states, such as free radicals and divalent molecules. Hydrocarbons adsorbed on the surface can be excited by electron collisions in the plasma, providing an additional viable reaction path.
Encoding disk refers to a digital encoder that measures angular displacement. It has the advantages of strong resolving power, high measurement accuracy and reliable work. It is one of the most commonly used displacement sensors for measuring the position of the shaft rotation angle. The code disk is divided into two types: absolute encoder and incremental encoder. The former can directly give the digital code corresponding to the angular position; the latter uses a computing system to make the pulse increment generated by the rotating code disc against a certain reference number addition and subtraction. The metal encoding disk is directly engraved with through and unconnected lines, and it is not fragile, but because the metal is not easy to corrode but easy deform, the accuracy is limited, and its thermal stability is an order of magnitude worse than that of glass. It is mainly used in motor, hardware, electrical appliances, automobile and other fields.
We customize diverse patterns metal encoder disk with drawings provided by customers. We are equipped with professional metal etching equipment and exposure development equipment. The raw material use for encoder disk are SUS304 stainless steel and the flat degree can below 0.02 mm. Our stainless-steel encoder disk can achieve flat, no burr and can be available in harsh environment.
Stainless Steel Encoder Disk,Encoder Disk,Metal Encoding Disk
SHAOXING HUALI ELECTRONICS CO., LTD. , https://www.cnsxhuali.com
